What Is An Ionic Bond
What Is An Ionic Bond - The calendar for 2025 is a useful tool to stay on top of plans in 2025. With various formats including monthly and yearly views, it offers convenience for planning. Whether you're organizing life or professional deadlines, this calendar keeps everything in order with simplicity and structure.
You can select a range of aesthetic or functional templates to suit your needs. Simply access and print from the comfort of your workspace or home. A 2025 printable calendar is ideal for planning holidays, noting important events, and managing priorities all year long.
What Is An Ionic Bond
What Is An Ionic Bond
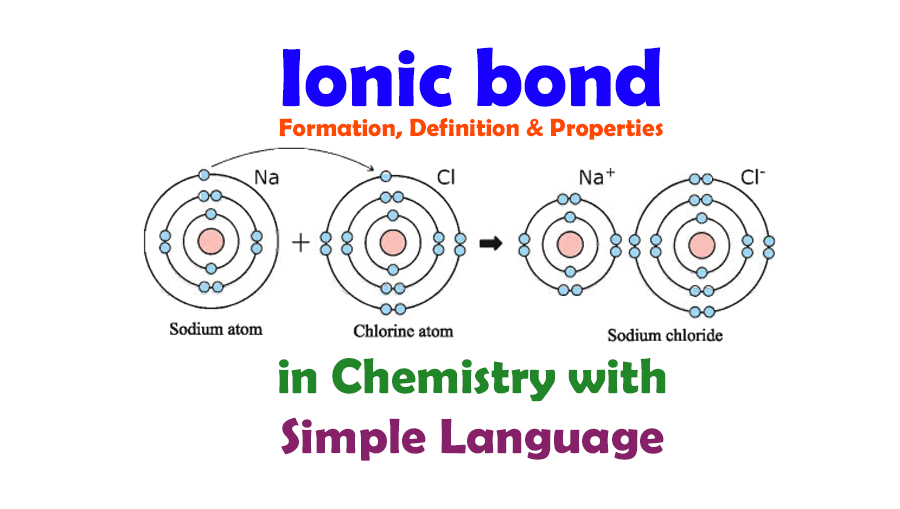
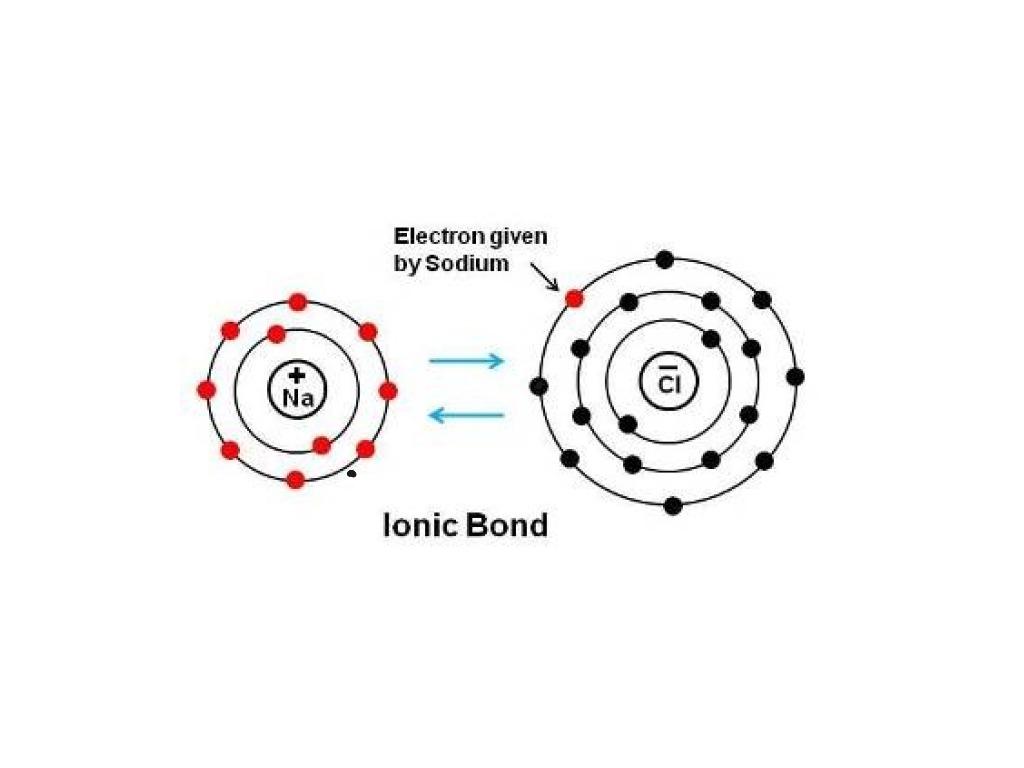
Ionic bonding is a form of chemical connection in which one atom loses valence electrons and gains them from another For both atoms involved this exchange results in a more stable noble gas electrical state The attractive electrostatic interactions between two ions of opposite charge form an ionic bond Q3 An ionic bond or electrovalent bond is an electrostatic attraction where one atom donates an electron to another atom. The transfer results in the atom that loses an electron become a positively charged ion or cation, while the atom gaining the electron becomes a negatively charged ion or anion.
8 2 Ionic Bonding Chemistry LibreTexts
What Is An Ionic Bond Sciencing Ionic Bonding Ionic Chemical Bond
What Is An Ionic BondCompounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. The properties of ionic compounds shed some light on the nature of ionic bonds. What is an Ionic Bond An ionic bond also known as an electrovalent bond is a type of chemical bond formed due to the electrostatic attraction between oppositely charged ions in a compound or molecule Ionic bond forms when the valence outermost electrons of one atom are transferred permanently to another atom following the octet rule
One type of chemical bond is an ionic bond. Ionic bonds result from the attraction between oppositely charged ions. For example, sodium cations (positively charged ions) and chlorine anions (negatively charged ions) are connected via ionic bonds in. Ionic Bond Definition Properties Formation Examples Applications Ionic Bond Definition Types Properties Examples
Ionic Bond Definition And Examples Science Notes And Projects

Ionic Bond Definition Types Properties Examples
Ionic bonding is a type of chemical bond in which valence electrons are lost from one atom and gained by another This exchange results in a more stable noble gas electronic configuration for both atoms involved An ionic bond is based on attractive electrostatic forces between two ions of opposite charge Cations and Anions Classify The Following Compounds As Having Covalent Or Ionic Bonds 42
Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions or between two atoms with sharply different electronegativities and is the primary interaction occurring in ionic compounds Ionic Bond Examples Biology Dictionary Fourth Grade GC August 2013

Ionic Bond Definition Examples Formation Chemical Bonding

Does Ion Bonding Change The Color Of Stainless Sekaautos
/ionic-bond-58fd4ea73df78ca1590682ad.jpg)
Examples Of Ionic Bonds And Ionic Compounds

Ionic Bond Definition Properties Examples Facts Britannica

Ionic Bond
Examples Of Ionic Bonds And Compounds

Ionic Bonding Wikipedia
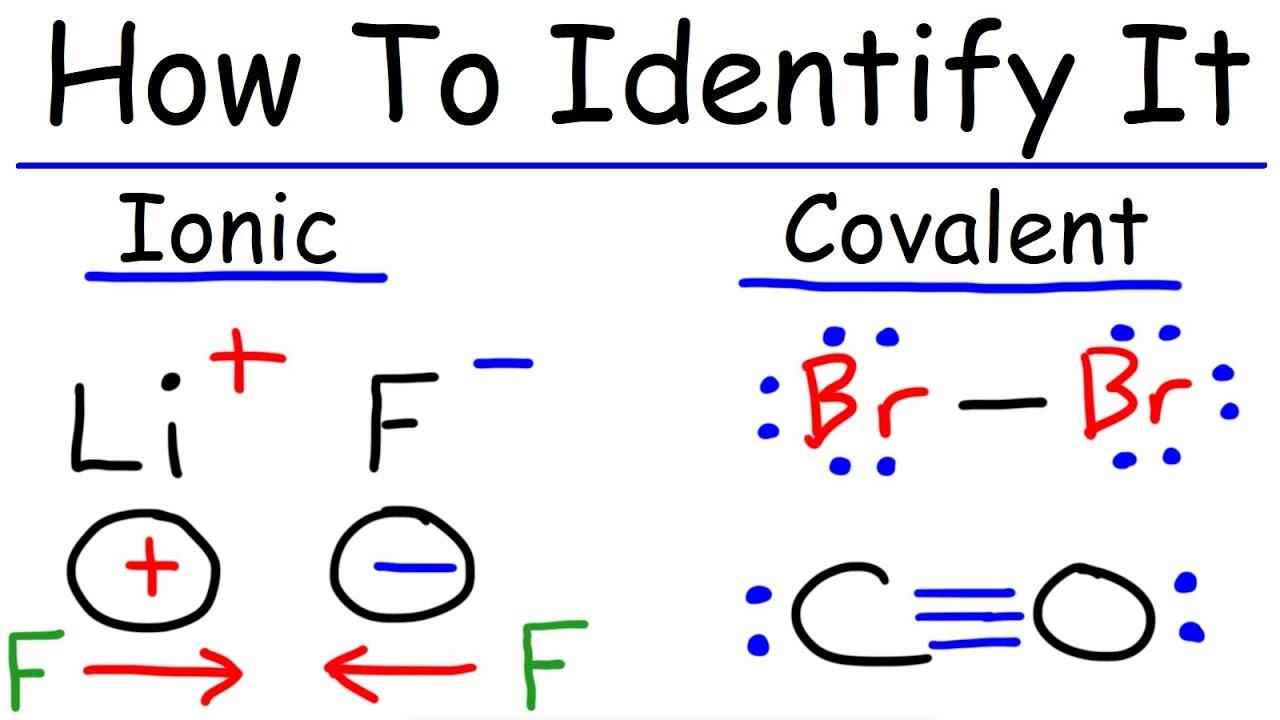
Classify The Following Compounds As Having Covalent Or Ionic Bonds 42
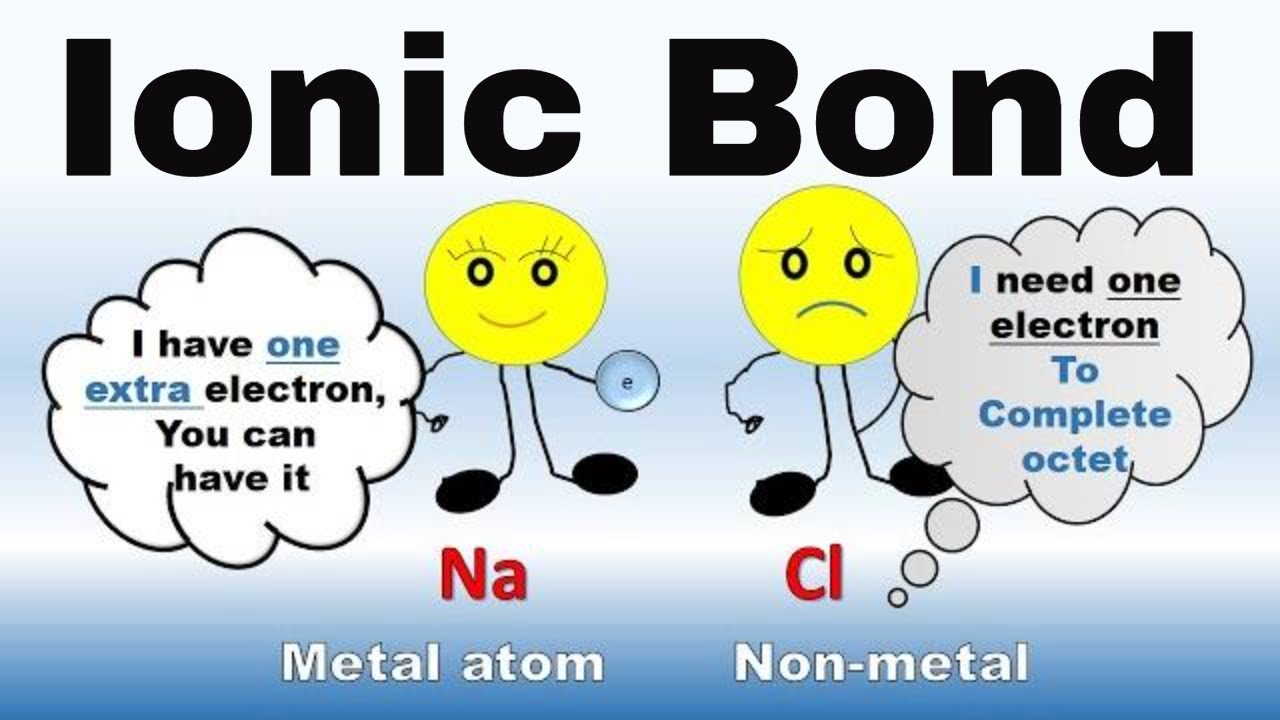
What Are Ionic Bonds Definition And Examples How Is An Ionic Bond
Ionic Bond Science Chemistry Chemical Bonds ShowMe
.PNG)