Alcoa Data Integrity Fda
Alcoa Data Integrity Fda - The calendar for 2025 is a handy tool to stay on top of plans in 2025. With multiple layout options, it offers convenience for planning. Whether you're tracking goals or business tasks, this calendar makes planning simple with simplicity and structure.
You can select a range of aesthetic or functional templates to suit your needs. Simply get it and start using from the comfort of your workspace or home. A 2025 printable calendar is perfect for planning holidays, noting important events, and staying productive throughout 2025.
Alcoa Data Integrity Fda

Alcoa Data Integrity Fda
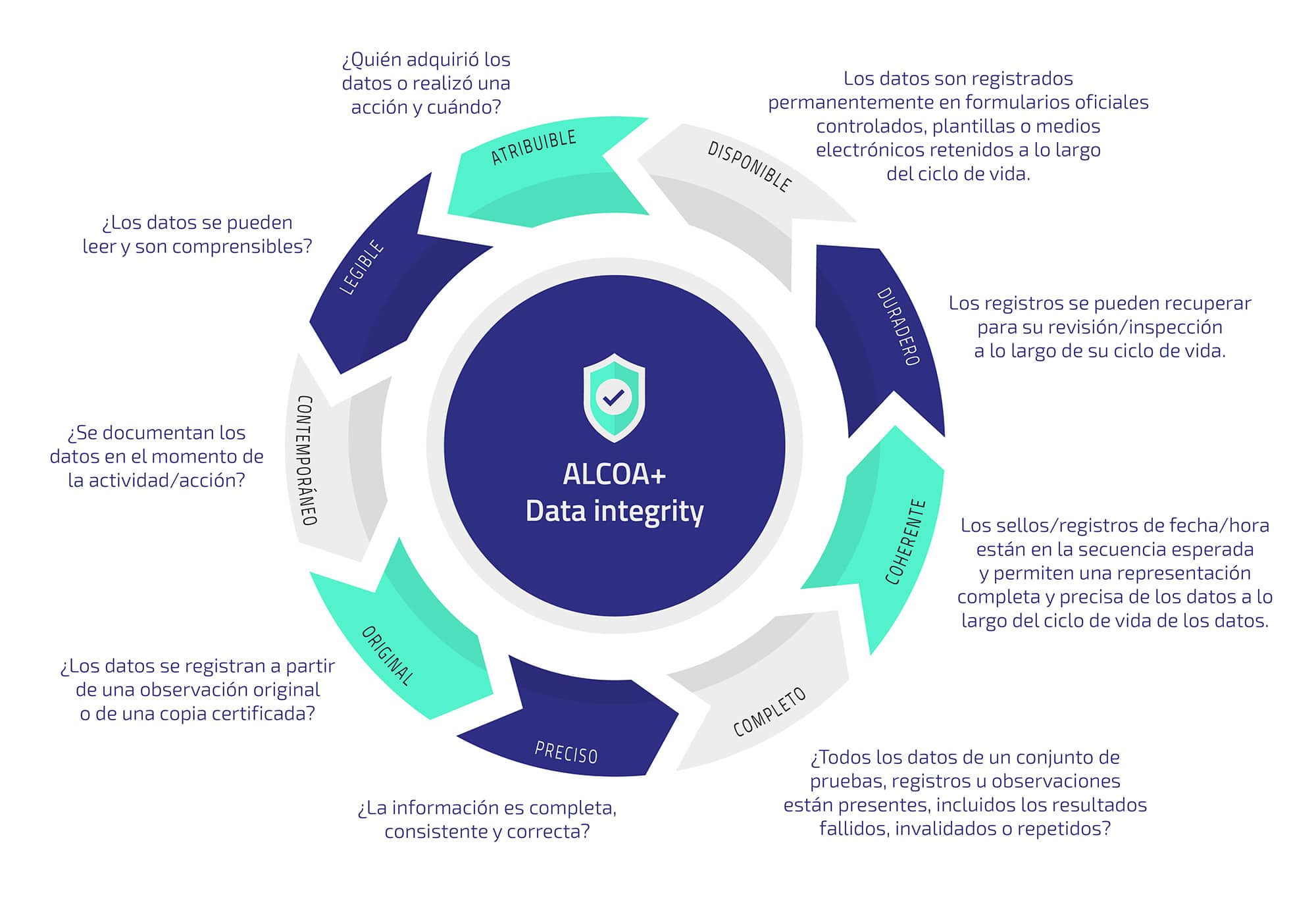
Az ALCOA az ALCOA 233 s az ALCOA dokumentumban le 237 rt alapelvek az adatintegrit 225 s el 233 r 233 s 233 re ir 225 nyul 243 t 246 rekv 233 seket tartalmazz 225 k valamint biztos 237 tj 225 k t 246 bbek k 246 z 246 tt az adatok The acronym ALCOA stands for Attributable, Legible, Contemporaneous, Original, Accurate. It has been reported, that the term was introduced by Stan W. Woollen (Senior Compliance …
Alcoa Wikipedia

ALCOA And ALCOA In Pharmaceuticals Principles Of ALCOA Data
Alcoa Data Integrity FdaJun 27, 2024 · ALCOA, an acronym standing for Attributable, Legible, Contemporaneous, Original, and Accurate, has long served as a cornerstone for data integrity. However, with … May 2 2024 nbsp 0183 32 The term ALCOA is an Acronym which stands for Attributable Legible Contemporaneous Original and Accurate ALCOA was then expanded to ALCOA Plus
Hogyan kell az Alcoa Dura-Bright ® kerekeket tisztítani? Videó a Dura-Bright ® kerekek tisztításáról ALCOA MES Engineer ALCOA Principles In Life Sciences And Pharmaceutical Production TotalLab
ALCOA What s New What s Important And What You Need To

What Is Data Integrity And ALCOA Principles I New ALCOA Plus Criteria I
Alcoa has the industry s most extensive portfolio of low carbon products in our Sustana brand With active operations across the full upstream aluminum industry we take a comprehensive DATA INTEGRITY Foundation Of Compliance Critical Source Of FDA
Ensure data integrity through ALCOA Plus As defined by FDA guidance to meet regulatory requirements your laboratory data must be attributable legible contemporaneous original DATA INTEGRITY Foundation Of Compliance Critical Source Of FDA ALCOA ALCOA Data Integrity

The Essential Guide To Data Integrity For Alcoa And Alcoa YouTube

Data Integrity 86

A Complete Guide To Data Integrity And ALCOA Principles In 44 OFF
A Complete Guide To Data Integrity And ALCOA Principles In 44 OFF

ALCOA C Qolty Medical Research Clinical Research Clinic

Ensuring Data Integrity CI Precision

Data Integrity Principle ALCOA To ALCOA Data Integrity Principles

DATA INTEGRITY Foundation Of Compliance Critical Source Of FDA
Data Integrity Asfalia

Period Net Download Starting Mutual Of That Book Are Proscribed